Developmental And Reproductive Toxicology (DART) Studies: How Do They Fit Into Your Program? | Premier Consulting

Immunoaffinity nanogold coupled with direct analysis in real time (DART) mass spectrometry for analytical toxicology - Analytical Methods (RSC Publishing)

Considerations in assessing the developmental and reproductive toxicity potential of biopharmaceuticals - Martin - 2009 - Birth Defects Research Part B: Developmental and Reproductive Toxicology - Wiley Online Library
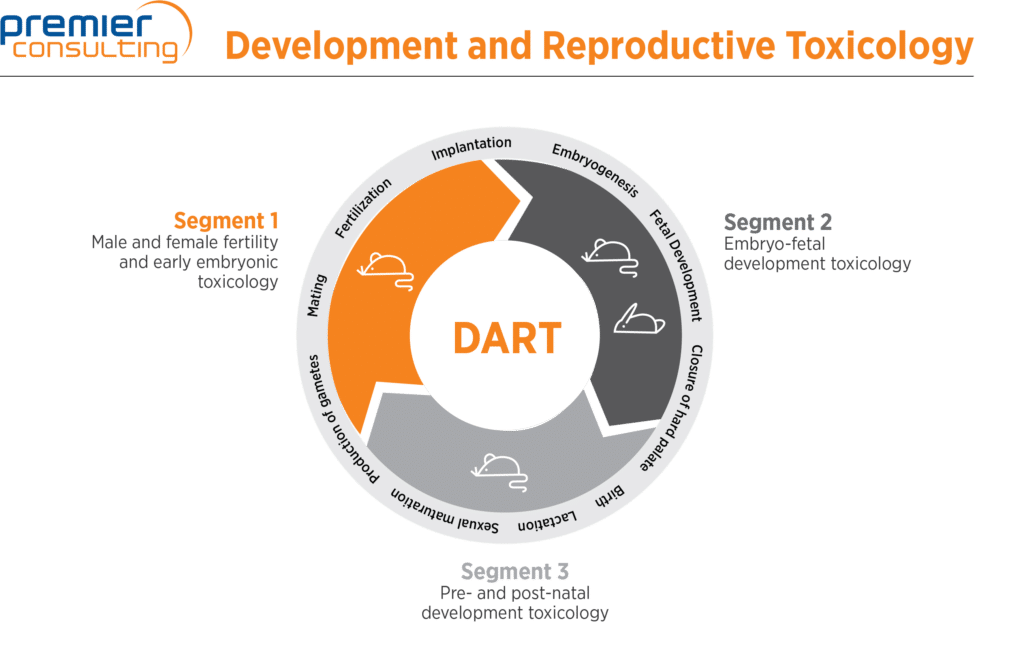
Developmental and Reproductive Toxicology (DART) - HESI - Health and Environmental Sciences Institute
![PDF] DarT: The embryo test with the Zebrafish Danio rerio--a general model in ecotoxicology and toxicology. | Semantic Scholar PDF] DarT: The embryo test with the Zebrafish Danio rerio--a general model in ecotoxicology and toxicology. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3002c3dbed270179655685d79b512eb80e1a2458/3-Figure1-1.png)
PDF] DarT: The embryo test with the Zebrafish Danio rerio--a general model in ecotoxicology and toxicology. | Semantic Scholar

ELSI & DART 10/1/04 Ethical, Legal, and Social Issues Related to DART ELSI – Children & Toxicology Reproductive & Developmental Toxicology: Pharmaceutical, - ppt download

DART - "Developmental And Reproductive Toxicology (database / base de données)" by AcronymsAndSlang.com

FDA Guidelines For Developmental and Reproductive Toxicology (DART) Studies for Small Molecules | Leaders in Pharmaceutical Business Intelligence (LPBI) Group

Carcinogenicity *General Toxicology **DART **Safety Pharmacology HighMedium Low Genetic Toxicity Pharmacokinetics Pharmacology Study Group Priority Study. - ppt download